Acinetobacter Baumannii
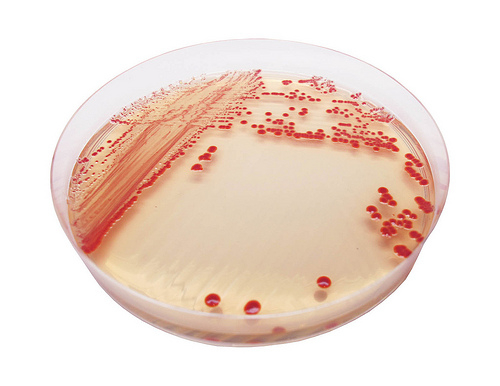
Acinetobacters are environmental organisms that are widespread both in and outside healthcare premises. The main species associated with human infection is Acinetobacter baumannii. In general, patients are more likely to be colonised rather than infected with A. baumannii. Susceptible patients are usually immunosuppressed or seriously ill due to other causes. Acinetobacters can occasionally cause infection in hospital patients- especially those who are already very ill, such as intensive care unit patients. It should be noted that the number of patients with serious bloodstream infections due to A. baumannii is much lower than Staphylococcus aureus (MRSA). In the UK, the A. baumannii organism is not often a cause of death, but may complicate therapy. There have also been recent reports of more virulent strains circulating in France. The Health Protection Agency is planning to review the situation in English acute NHS Trusts early in 2004 and is also preparing interim guidance on the control of the multi-resistant acinetobacter (see Health Protection Agency report).
What causes the spread of these organisms?
Are they virulent?
Is this a superbug?
So are they a problem?
Integrons: Integrons are chains of genes wherein many of the resistances acquired by the A. baumannii bacteria are found. The pathogen also has other options for their acquisition but it is the integrons that provide the most efficacious way to acquire and transmit the resistances, given that, apart from acquiring resistances, integrons have great mobility and can transfer from one location of the A. baumannii chromosome to another.
This mobility allows them to pass from one strain of the bacteria to another. This means that all the resistances acquired by a strain of A. baumannii can be transmitted to another and the species can thus modify and regenerate itself continuously. Moreover, as it has a promotoros, the bacteria is always activating or expressing all the resistances held in the integron.
Attempting to improve control
Analysing and isolating a number of A. baumannii strains from hospitals, it has been shown that most have integrons. Thus, it is highly probable that A. baumannii becomes resistant to the best antibiotics that exist today and that this resistance is transmitted via integrons. Moreover, A. baumannii strains have been identified that are resistant to the most common antibiotics used today.
If this is confirmed, the mortality rate due to infections created by the bacteria may even be greater than thought to date, given that there is no antibiotic capable of tackling the infection. It should be taken into account that the number of hospital patients affected by infections caused by A. baumannii is not great, but the gravity of the problem lies in the rate of mortality of these cases.
There currently exist methods to genetically distinguish A. baumannii strains from each other, but the aim at the moment is to obtain a method of indicating the presence of integrons and their resistance in these strains. Of course, this method of detection has to be standardised and, at the same time, practical, for its clinical use.
That is to say, the option of the researchers has been to try to improve control with respect to A. baumannii given that there is currently no substitutes for the antibiotics used to date. In order to achieve this improved control, it is essential to detect the infection in time and know if A. baumannii has produced it. The resistances of the strains must also be known and if they have integrons. Once this detailed information is gathered, new systems for the control of infections can be introduced in order keep down the rate of mortality due to A. baumannii. They may respond to Colloidal Silver and Colloidal Copper.
Heart-breaking bugs. Infections trigger heart disease?
Debora MacKenzie
“Nearly five million chickens quarantined in bird flu scare” at http://www.cnn.com/2003/US/Northeast/03/06/chicken.quarantine.ap/index.html


































Get Social